Human Cord Blood CD19+ B Cells
B cells, also known as B lymphocytes, express CD19 from very early stages, and the expression is only lost upon maturation into plasma cells. After maturing, the B cells migrate to secondary lymphatic organs and are subsequently activated directly by an antigen or through an antigen presenting cell (APS). At this point they either become memory B cells or plasma B cells. Each B cell gives rise to a single species of antibody.
Our Human Cord Blood CD19+ B cells are isolated from cord blood mononuclear cells by positive immunomagnetic selection.
All cord blood is collected in Citric Phosphate with Dextrose Buffer (CPD) from fully consented IRB approved donors who are tested negative for HIV, HBV and HCV.

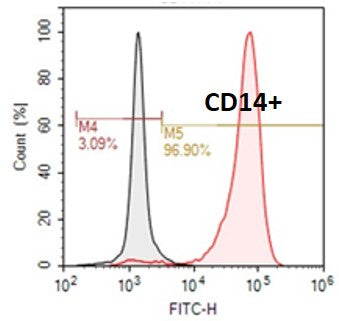
Figure 1. Flow cytometric analysis showed that >95% cells are CD19 positive.
CD19
CD19 (Cluster of Differentiation 19), is a protein that is found on the surface of B-cells (B lymphocyte). So it is also known as B lymphocyte antigen.
Function
Lymphocytes proliferate and differentiate in response to various concentrations of different antigens. The ability of the B cell to respond in a specific, yet sensitive manner to the various antigens is achieved with the use of low-affinity antigen receptors. The CD19 gene encodes a cell surface molecule that assembles with the antigen receptor of B lymphocytes in order to decrease the threshold for antigen receptor-dependent stimulation.
CD19 is expressed on follicular dendritic cells and B cells. In fact, it is present on B cells from earliest recognizable B-lineage cells during development to B-cell blasts but is lost on maturation to plasma cells. It primarily acts as a B cell co-receptor in conjunction with CD21 and CD81. Upon activation, the cytoplasmic tail of CD19 becomes phosphorylated, which leads to binding by Src-family kinases and recruitment of PI-3 kinase.
As on T cells, several surface molecules form the antigen receptor and form a complex on B lymphocytes. The (almost) B cell-specific CD19 phosphoglycoprotein is one of these molecules. The others are CD21 and CD81. These surface immunoglobulin (sIg)-associated molecules facilitate signal transduction. On B cells, anti-immunoglobulin antibody mimicking exogenous antigen causes CD19 to bind to sIg and internalize with it. The reverse process has not been demonstrated, suggesting that formation of this receptor complex is antigen-induced. This molecular association has been confirmed by chemical studies.

BCR indicates B-cell receptor; BTK, Bruton tyrosine kinase.
Source: GeneCards, www.genecards.
In disease
Mutations in CD19 are associated with severe immunodeficiency syndromes characterized by diminished antibody production.
Since CD19 is a hallmark of B-cells, the protein has been used to diagnose cancers that arise from this type of cell - notably B-cell lymphomas. Since 2011, anti-CD19 immunotoxin treatments targeting CD19 have begun to enter trials. Most current experimental anti-CD19 drugs in development work by exploiting the presence of CD19 to direct treatment specifically towards B-cell cancers. However, it is now emerging that the protein plays an active role in driving the growth of these cancers, most intriguingly by stabilizing the concentrations of the MYC oncoprotein. This suggests that CD19 and its downstream signaling may be a more attractive therapeutic target than suspected.
CD19 has also been implicated in autoimmune diseases and may be a useful treatment target.